Chemistry:Quetiapine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /kwɪˈtaɪ.əpiːn/ kwi-TY-ə-peen |
| Trade names | Seroquel, Seroquel XR, Temprolide, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698019 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Atypical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100%[1] |
| Protein binding | 83%[2] |
| Metabolism | Liver via CYP3A4-catalysed sulfoxidation to its active metabolite norquetiapine (N-desalkylquetiapine)[5] |
| Elimination half-life | 7 hours (parent compound); 9–12 hours (active metabolite, norquetiapine)[2][3] |
| Excretion | Kidney (73%), feces (20%)[1][2][3][4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
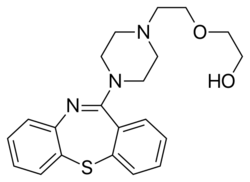
| Formula | C21H25N3O2S |
| Molar mass | 383.51 g·mol−1 |
| 3D model (JSmol) | |
| Solubility in water | 3.29 mg/mL (20 °C) |
| |
| |
| (verify) | |
Quetiapine, sold under the brand name Seroquel among others, is an atypical antipsychotic medication used for the treatment of schizophrenia, bipolar disorder, and major depressive disorder.[6][7] Despite being widely used as a sleep aid due to its sedating effect, the benefits of such use do not appear to generally outweigh the side effects.[8] It is taken orally.[6]
Common side effects include sleepiness, constipation, weight gain, and dry mouth.[6] Other side effects include low blood pressure with standing, seizures, a prolonged erection, high blood sugar, tardive dyskinesia, and neuroleptic malignant syndrome.[6] In older people with dementia, its use increases the risk of death.[6] Use in the third trimester of pregnancy may result in a movement disorder in the baby for some time after birth.[6] Quetiapine is believed to work by blocking a number of receptors, including those for serotonin and dopamine.[6]
Quetiapine was developed in 1985 and approved for medical use in the United States in 1997.[6][9] It is available as a generic medication.[10] In 2021, it was the 62nd most commonly prescribed medication in the United States, with more than 10 million prescriptions.[11][12] It is on the World Health Organization's List of Essential Medicines.[13]
Medical uses
Quetiapine is primarily used to treat schizophrenia or bipolar disorder.[14] Quetiapine targets both positive and secondary negative symptoms of schizophrenia.[15]
Schizophrenia
A 2013 Cochrane review compared quetiapine to typical antipsychotics:
| Summary | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quetiapine may not differ from typical antipsychotics in the treatment of positive symptoms, general psychopathology, and negative symptoms. However, it causes fewer adverse effects in terms of abnormal ECG, extrapyramidal effects, abnormal prolactin levels and weight gain.[16] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In a 2013 comparison of 15 antipsychotics in effectiveness in treating schizophrenia, quetiapine demonstrated standard effectiveness. It was 13–16% more effective than ziprasidone, chlorpromazine, and asenapine and approximately as effective as haloperidol and aripiprazole.[17]
There is tentative evidence of the benefit of quetiapine versus placebo in schizophrenia; however, definitive conclusions are not possible due to the high rate of attrition in trials (greater than 50%) and the lack of data on economic outcomes, social functioning, or quality of life.[18]
It is debatable whether, as a class, typical or atypical antipsychotics are more effective.[19] Both have equal drop-out and symptom relapse rates when typicals are used at low to moderate dosages.[20] While quetiapine has lower rates of extrapyramidal side effects, there is greater sleepiness and rates of dry mouth.[18]
A Cochrane review comparing quetiapine to other atypical antipsychotic agents tentatively concluded that it may be less efficacious than olanzapine and risperidone; produce fewer movement related side effects than paliperidone, aripiprazole, ziprasidone, risperidone and olanzapine; and produce weight gain similar to risperidone, clozapine and aripiprazole. They concluded that it produces suicide attempt, suicide; death; QTc prolongation, low blood pressure; tachycardia; sedation; gynaecomastia; galactorrhoea, menstrual irregularity and white blood cell count at a rate similar to first generation antipsychotics.[21]
Bipolar disorder
In those with bipolar disorder, quetiapine is used to treat depressive episodes; acute manic episodes associated with bipolar I disorder (as either monotherapy or adjunct therapy to lithium; valproate or lamotrigine); acute mixed episodes; and maintenance treatment of bipolar I disorder (as adjunct therapy to lithium or divalproex).
Major depressive disorder
Quetiapine is effective when used by itself[7] and when used along with other medications in major depressive disorder (MDD).[7][22] However, sedation is often an undesirable side effect.[7]
In the United States,[3] the United Kingdom[23] and Australia (while not subsidised by the Australian Pharmaceutical Benefits Scheme for treatment of MDD), quetiapine is licensed for use as an add-on treatment in MDD.[24]
Alzheimer's disease
Quetiapine does not decrease agitation among people with Alzheimer's disease. Quetiapine worsens intellectual functioning in the elderly with dementia and therefore is not recommended.[25]
Others
The use of low doses of quetiapine for insomnia, while common, is not recommended; there is little evidence of benefit and concerns regarding adverse effects.[26][27][28][29][30][31] A 2022 network meta-analysis of 154 double-blind, randomized controlled trials of drug therapies vs. placebo for insomnia in adults found that quetiapine did not demonstrate any short-term benefits in sleep quality. Quetiapine, specifically, had an effect size (standardized mean difference) against placebo for treatment of insomnia of 0.05 (95% CI –1.21 to 1.11) at 4 weeks of treatment, with the certainty of evidence rated as very low.[32] Doses of quetiapine used for insomnia have ranged from 12.5 to 800 mg, with low doses of 25 to 200 mg being the most typical.[33][26][27] Regardless of the dose used, some of the more serious adverse effects may still possibly occur at the lower dosing ranges, such as dyslipidemia and neutropenia.[34][35] These safety concerns at low doses are corroborated by Danish observational studies that showed use of specifically low-dose quetiapine (prescriptions filled for tablet strengths >50 mg were excluded) was associated with an increased risk of major cardiovascular events as compared to use of Z-drugs, with most of the risk being driven by cardiovascular death.[36] Laboratory data from an unpublished analysis of the same cohort also support the lack of dose-dependency of metabolic side effects, as new use of low-dose quetiapine was associated with a risk of increased fasting triglycerides at 1-year follow-up.[37]
It is sometimes used off-label, often as an augmentation agent, to treat conditions such as Tourette syndrome,[38] musical hallucinations[39] and anxiety disorders.[40]
Quetiapine and clozapine are the most widely used medications for the treatment of Parkinson's disease psychosis due to their relatively low extrapyramidal side-effect liability.[41] Owing to the risks associated with clozapine (e.g. agranulocytosis, diabetes mellitus, etc.), clinicians often attempt treatment with quetiapine first, although the evidence to support quetiapine's use for this indication is significantly weaker than that of clozapine.[42][43]
Adverse effects
Sources for incidence lists:[1][3][23][24][43][44]
- Very common (>10% incidence) adverse effects
- Dry mouth
- Dizziness
- Headache
- Somnolence (drowsiness; of 15 antipsychotics quetiapine causes the 5th most sedation. Extended release (XR) formulations tend to produce less sedation, dose-by-dose, than the immediate release formulations.)[17]
- Common (1–10% incidence) adverse effects
- High blood pressure
- Orthostatic hypotension
- High pulse rate
- High blood cholesterol
- Elevated serum triglycerides
- Abdominal pain
- Constipation
- Increased appetite
- Vomiting
- Increased liver enzymes
- Backache
- Asthenia
- Insomnia
- Lethargy
- Tremor
- Agitation
- Nasal congestion
- Pharyngitis
- Fatigue
- Pain
- Dyspepsia (Indigestion)
- Peripheral oedema
- Dysphagia
- Extrapyramidal disease: Quetiapine and clozapine are noted for their relative lack of extrapyramidal side effects.[17][23][43]
- Weight gain: SMD 0.43 kg when compared to placebo. Produces roughly as much weight gain as risperidone, less weight gain than clozapine, olanzapine and zotepine and more weight gain than ziprasidone, lurasidone, aripiprazole and asenapine.[17] As with many other atypical antipsychotics, this action is likely due to its actions at the H1 histamine receptor and 5-HT2C receptor.[5]
- Rare (<1% incidence) adverse effects
- Prolonged QT interval (had an odds ratio for prolonging the QT interval over placebo of 0.17)[17]
- Sudden cardiac death
- Syncope
- Diabetic ketoacidosis
- Restless legs syndrome
- Hyponatraemia, low blood sodium.
- Jaundice, yellowing of the eyes, skin and mucous membranes due to an impaired ability of the body to clear bilirubin, a by product of haem breakdown.
- Pancreatitis, pancreas swelling.
- Agranulocytosis, a potentially fatal drop in white blood cell count.
- Leukopenia, a drop in white blood cell count, not as severe as agranulocytosis.
- Neutropenia, a drop in neutrophils, the cell of the immune cells that defends the body against bacterial infections.
- Eosinophilia
- Anaphylaxis, a potentially fatal allergic reaction.
- Seizure
- Hypothyroidism, underactive thyroid gland.
- Myocarditis, swelling of the myocardium.
- Cardiomyopathy
- Hepatitis, swelling of the liver.
- Suicidal ideation
- Priapism. A prolonged and painful erection.
- Stevens–Johnson syndrome. A potentially fatal skin reaction.
- Neuroleptic malignant syndrome a rare and potentially fatal complication of antipsychotic drug treatment. It is characterised by the following symptoms: tremor, rigidity, hyperthermia, tachycardia, mental status changes (e.g. confusion), etc.
- Tardive dyskinesia. A rare and often irreversible neurological condition characterised by involuntary movements of the face, tongue, lips and rest of the body. Most commonly occurs after prolonged treatment with antipsychotics. It is believed to be particularly uncommon with atypical antipsychotics, especially quetiapine and clozapine[24]
Both typical and atypical antipsychotics can cause tardive dyskinesia.[45] According to one study, rates are lower with the atypicals at 3.9% as opposed to the typicals at 5.5%.[45] Although quetiapine and clozapine are atypical antipsychotics, switching to these atypicals is an option to minimize symptoms of tardive dyskinesia caused by other atypicals.[46]
Weight gain can be a problem for some, with quetiapine causing more weight gain than fluphenazine, haloperidol, loxapine, molindone, olanzapine, pimozide, risperidone, thioridazine, thiothixene, trifluoperazine, and ziprasidone, but less than chlorpromazine, clozapine, perphenazine, and sertindole.[47]
As with some other anti-psychotics, quetiapine may lower the seizure threshold,[48] and should be taken with caution in combination with drugs such as bupropion.
Discontinuation
The British National Formulary recommends a gradual withdrawal when discontinuing antipsychotics to avoid acute withdrawal syndrome or rapid relapse.[49] Symptoms of withdrawal commonly include nausea, vomiting, and loss of appetite.[50] Other symptoms may include restlessness, increased sweating, and trouble sleeping.[50] Less commonly there may be a feeling of the world spinning, numbness, or muscle pains.[50] Symptoms generally resolve after a short period of time.[50]
There is tentative evidence that discontinuation of antipsychotics can result in psychosis.[51] It may also result in reoccurrence of the condition that is being treated.[52] Rarely tardive dyskinesia can occur when the medication is stopped.[50]
Pregnancy and lactation
Placental exposure is least for quetiapine compared to other atypical antipsychotics.[43] The evidence is insufficient to rule out any risk to the foetus but available data suggests it is unlikely to result in any major foetal malformations.[2][4][44] It is secreted in breast milk and hence quetiapine-treated mothers are advised not to breastfeed.[2][4][44]
Abuse potential
In contrast to most other antipsychotic drugs, which tend to be somewhat aversive and often show problems with patient compliance with prescribed medication regimes, quetiapine is sometimes associated with drug misuse and abuse potential, for its hypnotic and sedative effects. It has a limited potential for misuse, usually only in individuals with a history of polysubstance abuse and/or mental illness, and especially in those incarcerated in prisons or secure psychiatric facilities where access to alternative intoxicants is more limited. To a significantly greater extent than other atypical antipsychotic drugs, quetiapine was found to be associated with drug-seeking behaviors, and to have standardised street prices and slang terms associated with it, either by itself or in combination with other drugs (such as "Q-ball" for the intravenous injection of quetiapine mixed with cocaine). The pharmacological basis for this distinction from other second generation antipsychotic drugs is unclear, though it has been suggested that quetiapine's comparatively lower dopamine receptor affinity and strong antihistamine activity might mean it could be regarded as more similar to sedating antihistamines in this context. While these issues have not been regarded as sufficient cause for placing quetiapine under increased legal controls, prescribers have been urged to show caution when prescribing quetiapine to individuals with characteristics that might place them at increased risk for drug misuse.[53][54][55][56][57]
Overdose
Most instances of acute overdosage result in only sedation, hypotension and tachycardia, but cardiac arrhythmia, coma and death have occurred in adults. Serum or plasma quetiapine concentrations are usually in the 1–10 mg/L range in overdose survivors, while postmortem blood levels of 10–25 mg/L are generally observed in fatal cases.[58] Non-toxic levels in postmortem blood extend to around 0.8 mg/kg, but toxic levels in postmortem blood can begin at 0.35 mg/kg.[59][60]
Pharmacology
Pharmacodynamics
| Site | QTP | NQTP | Action | Ref |
|---|---|---|---|---|
| NET | >10,000 | 58 | Blocker | [62] |
| DAT | >10,000 | >10,000 | ND | [62] |
| 5-HT1A | 320–432 | 45 | Partial agonist | [62][63] |
| 5-HT1B | 1,109–2,050 | 1,117 | ND | [62][63] |
| 5-HT1D | >10,000 | 249 | ND | [62][63] |
| 5-HT1E | 1,250–2,402 | 97 | ND | [62][63] |
| 5-HT1F | 2,240 | ND | ND | [63] |
| 5-HT2A | 96–101 | 48 | Antagonist | [62][63] |
| 5-HT2B | ND | 14 | Antagonist | [62] |
| 5-HT2C | 2,502 | 107 | Antagonist | [62] |
| 5-HT3 | >10,000 | 394 | Antagonist | [62] |
| 5-HT4 | ND | ND | ND | ND |
| 5-HT5A | 3,120 | 768 | ND | [62] |
| 5-HT6 | 1,865 | 503 | Antagonist | [62] |
| 5-HT7 | 307 | 76 | Antagonist | [62] |
| α1A | 22 | 144 | Antagonist | [62] |
| α1B | 39 | 95 | Antagonist | [62] |
| α2A | 2,230–3,630 | 237 | Antagonist | [62][63] |
| α2B | 90–747 | 378 | Antagonist | [62][63] |
| α2C | 28.7–350 | 736 | Antagonist | [62][63] |
| β1 | >10,000 | >10,000 | ND | [62][63] |
| β2 | >10,000 | >10,000 | ND | [62][63] |
| D1 | 712 | 214 | Antagonist | [62] |
| D2 | 245 | 196 | Antagonist | [62] |
| D2L | 700 | ND | Antagonist | [63] |
| D2S | 390 | ND | Antagonist | [63] |
| D3 | 340–483 | 567 | Antagonist | [62][63] |
| D4 | 1,202 | 1,297 | Antagonist | [62] |
| D4.2 | 1,600 | ND | Antagonist | [63] |
| D5 | 1,738 | 1,419 | Antagonist | [62] |
| H1 | 2.2–11 | 3.5 | Antagonist | [62][63] |
| H2 | >10,000 | 298 | Antagonist | [62] |
| H3 | >10,000 | >10,000 | ND | [62] |
| H4 | >10,000 | 1,660 | ND | [62] |
| M1 | 858 | 39 | Antagonist | [62] |
| M2 | 1,339 | 453 | ND | [62] |
| M3 | >10,000 | 23 | Antagonist | [62] |
| M4 | 542 | 110 | ND | [62] |
| M5 | 1,942 | 23 | Antagonist | [62] |
| σ1 | 220–3,651 | >10,000 | ND | [62][63] |
| σ2 | 1,344 | 1,050 | ND | [62] |
| NMDA (PCP) |
>10,000 | ND | Antagonist | [62] |
| VDCC | >10,000 | ND | ND | [62][63] |
| hERG | ND | >10,000 (IC50) |
ND | [62] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. All data are for human cloned proteins, except σ1 (guinea pig), σ2 (rat), and VDCC (rat).[62][63] | ||||
Quetiapine has the following pharmacological actions:[64][65][66][67][68][69][70][71]
- Dopamine D1, D2, D3, D4, and D5 receptor antagonist
- Serotonin 5-HT1A receptor partial agonist, 5-HT2A, 5-HT2B, 5-HT2C, 5-HT3, 5-HT6, and 5-HT7 receptor antagonist, and 5-HT1B, 5-HT1D, 5-HT1E, and 5-HT1F receptor ligand
- α1- and α2-adrenergic receptor antagonist
- Histamine H1 receptor antagonist
- Muscarinic acetylcholine receptor antagonist
This means quetiapine is a dopamine, serotonin, and adrenergic antagonist, and a potent antihistamine with some anticholinergic properties.[72] Quetiapine binds strongly to serotonin receptors; the drug acts as partial agonist at 5-HT1A receptors.[73] Serial PET scans evaluating the D2 receptor occupancy of quetiapine have demonstrated that quetiapine very rapidly disassociates from the D2 receptor.[74] Theoretically, this allows for normal physiological surges of dopamine to elicit normal effects in areas such as the nigrostriatal and tuberoinfundibular pathways, thus minimizing the risk of side-effects such as pseudo-parkinsonism as well as elevations in prolactin.[75] Some of the antagonized receptors (serotonin, norepinephrine) are actually autoreceptors whose blockade tends to increase the release of neurotransmitters.
At very low doses, quetiapine acts primarily as a histamine receptor blocker (antihistamine) and α1-adrenergic blocker. When the dose is increased, quetiapine activates the adrenergic system and binds strongly to serotonin receptors and autoreceptors. At high doses, quetiapine starts blocking significant amounts of dopamine receptors.[65][76] Due to the drug's sedating H1 activity, it is often prescribed at low doses for insomnia. While some feel that low doses of drugs with antihistamine effects like quetiapine and mirtazapine are safer than drugs associated with physical dependency or other risk factors, concern has been raised by some professionals that off-label prescribing has become too widespread due to underappreciated hazards.[77]
When treating schizophrenia, antagonism of D2 receptor by quetiapine in the mesolimbic pathway relieves positive symptoms and antagonism of the 5-HT2A receptor in the frontal cortex of the brain may relieve negative symptoms and reduce severity of psychotic episodes.[15][78][79] Quetiapine has fewer extrapyramidal side effects and is less likely to cause hyperprolactinemia when compared to other drugs used to treat schizophrenia, so is used as a first line treatment.[80][81]
Pharmacokinetics
Peak levels of quetiapine occur 1.5 hours after a dose.[82] The plasma protein binding of quetiapine is 83%.[82] The major active metabolite of quetiapine is norquetiapine (N-desalkylquetiapine).[62] Quetiapine has an elimination half-life of 6 or 7 hours.[82][2][3] Its metabolite, norquetiapine, has a half-life of 9 to 12 hours.[2][3] Quetiapine is excreted primarily via the kidneys (73%) and in feces (20%) after hepatic metabolism, the remainder (1%) is excreted as the drug in its unmetabolized form.[78][82]
Chemistry
Quetiapine is a tetracyclic compound and is closely related structurally to clozapine, olanzapine, loxapine, and other tetracyclic antipsychotics.
Synthesis
The synthesis of quetiapine begins with a dibenzothiazepinone. The lactam is first treated with phosphoryl chloride to produce a dibenzothiazepine. A nucleophilic substitution is used to introduce the sidechain.[83]
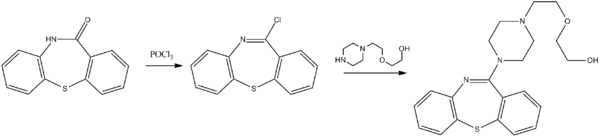
|
History
Sustained-release
AstraZeneca submitted a new drug application for a sustained-release version of quetiapine in the United States, Canada, and the European Union in the second half of 2006 for treatment of schizophrenia.[84][85] AstraZeneca was to retain the exclusive right to market sustained-release quetiapine until 2017. The sustained-release quetiapine is marketed mainly as Seroquel XR. Other marketing names are Seroquel Prolong, Seroquel Depot and Seroquel XL
On 18 May 2007, AstraZeneca announced that the U.S. FDA approved Seroquel XR for acute treatment of schizophrenia.[86] During its 2007 Q2 earnings conference, AstraZeneca announced plans to launch Seroquel XR in the U.S. during August 2007.[87] However, Seroquel XR has become available in U.S. pharmacies only after the FDA approved Seroquel XR for use as maintenance treatment for schizophrenia, in addition to acute treatment of the illness, on 16 November 2007.[88] The company has not provided a reason for the delay of Seroquel XR's launch.
Health Canada approved sale of Seroquel XR on 27 September 2007.[89]
In early October 2008, the FDA approved Seroquel XR for the treatment of bipolar depression and bipolar mania. According to AstraZeneca, Seroquel XR is "the first medication approved by the FDA for the once-daily acute treatment of both depressive and manic episodes associated with bipolar."
On 31 July 2008, Handa Pharmaceuticals, based in Fremont, California, announced that its abbreviated new drug application ("ANDA") for quetiapine fumarate extended-release tablets, the generic version of AstraZeneca's SEROQUEL XR, has been accepted by the FDA.
On 1 December 2008, Biovail announced that the FDA had accepted the company's ANDA to market its own version of sustained-release quetiapine.[90] Biovail's sustained-release tablets will compete with AstraZeneca's Seroquel XR.
On 24 December 2008, AstraZeneca notified shareholders that the FDA had asked for additional information on the company's application to expand the use of sustained-release quetiapine for treatment of depression.[91]
Society and culture
Regulatory status
In the United States, the Food and Drug Administration (FDA) has approved quetiapine for the treatment of schizophrenia and of acute manic episodes associated with bipolar disorder (bipolar mania) and for treatment of bipolar depression.[92] In 2009, quetiapine XR was approved as adjunctive treatment of major depressive disorder.[93]
Quetiapine received its initial indication from U.S. FDA for treatment of schizophrenia in 1997.[94] In 2004, it received its second indication for the treatment of mania-associated bipolar disorder.[95] In 2007 and 2008, studies were conducted on quetiapine's efficacy in treating generalized anxiety disorder and major depression.
Patent protection for the product ended in 2012; however, in a number of regions, the long-acting version remained under patent until 2017.[96]
Lawsuits
In April 2010, the U. S. Department of Justice fined Astra-Zeneca $520 million for the company's aggressive marketing of Seroquel for off-label uses.[92] According to the Department of Justice, "the company recruited doctors to serve as authors of articles that were ghostwritten by medical literature companies and about studies the doctors in question did not conduct. AstraZeneca then used those studies and articles as the basis for promotional messages about unapproved uses of Seroquel."[92]
Multiple lawsuits have been filed in relation to quetiapine's side-effects, in particular, diabetes.[97][98][99][100]
Approximately 10,000[101] lawsuits[102] have been filed against AstraZeneca, alleging that quetiapine caused problems ranging from slurred speech and chronic insomnia to deaths.
Controversy
In 2004, a young man named Dan Markingson committed suicide in a controversial Seroquel clinical trial at the University of Minnesota while under an involuntary commitment order.[103] A group of University of Minnesota bioethicists charged that the trial involved an alarming number of ethical violations.[104]
Nurofen Plus tampering case
In August 2011, the UK's Medicines and Healthcare products Regulatory Agency (MHRA) issued a class-4 drug alert following reports that some batches of Nurofen plus contained Seroquel XL tablets instead.[105]
Following the issue of the Class-4 Drug Alert, Reckitt Benckiser (UK) Ltd received further reports of rogue blister strips in cartons of two additional batches of Nurofen Plus tablets. One of the new batches contained Seroquel XL 50 mg tablets and one contained the Pfizer product Neurontin 100 mg capsules.
Following discussions with the MHRA's Defective Medicines Report Centre (DMRC), Reckitt Benckiser (UK) Ltd decided to recall all remaining unexpired stock of Nurofen Plus tablets in any pack size, leading to a Class-1 Drug Alert.[106] The contamination was later traced to in-store tampering by a customer.[107]
References
- ↑ 1.0 1.1 1.2 "quetiapine (Rx) - Seroquel, Seroquel XR". WebMD. http://reference.medscape.com/drug/seroquel-xr-quetiapine-342984#10.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 "Quetiapine 25 mg film-coated tablets - Summary of Product Characteristics". Sandoz. January 2013. http://www.medicines.org.uk/emc/medicine/26575/SPC/Quetiapine+25+mg+film-coated+tablets/.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Truven Health Analytics, Inc. DrugPoint System (Internet) [cited 2013 Sep 18]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ↑ 4.0 4.1 4.2 "Quetiapine fumarate tablet". Ascend Laboratories, LLC. October 2013. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=3112a006-1c61-47f2-84f5-9a7670d09c9b.
- ↑ 5.0 5.1 Goodman and Gilman's The Pharmacological Basis of Therapeutics (12th ed.). McGraw Hill Professional. 2010. ISBN 978-0071624428. https://books.google.com/books?id=bVUfAQAAQBAJ.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 "Quetiapine Fumarate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/quetiapine-fumarate.html.
- ↑ 7.0 7.1 7.2 7.3 "Second-generation antipsychotics for major depressive disorder and dysthymia". The Cochrane Database of Systematic Reviews (12): CD008121. December 2010. doi:10.1002/14651858.CD008121.pub2. PMID 21154393.
- ↑ "Quetiapine for insomnia: A review of the literature". American Journal of Health-System Pharmacy 71 (5): 394–402. March 2014. doi:10.2146/ajhp130221. PMID 24534594. https://pdfs.semanticscholar.org/8812/afd4bc713c03435b68f2bb6ea456c611b0b2.pdf.
- ↑ "Quetiapine in the treatment of schizophrenia and related disorders". Neuropsychiatric Disease and Treatment 3 (2): 219–235. April 2007. doi:10.2147/nedt.2007.3.2.219. PMID 19300555.
- ↑ British national formulary: BNF 74 (74 ed.). British Medical Association. 2017. p. 383. ISBN 978-0857112989.
- ↑ "The Top 300 of 2021". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Quetiapine - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Quetiapine.
- ↑ The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. 2023. WHO/MHP/HPS/EML/2023.02.
- ↑ "quetiapine-fumarate". https://www.drugs.com/monograph/quetiapine-fumarate.html.
- ↑ 15.0 15.1 "Quetiapine: a review of its safety in the management of schizophrenia". Drug Safety 23 (4): 295–307. October 2000. doi:10.2165/00002018-200023040-00003. PMID 11051217.
- ↑ 16.0 16.1 "Quetiapine versus typical antipsychotic medications for schizophrenia". The Cochrane Database of Systematic Reviews 5 (5): CD007815. May 2013. doi:10.1002/14651858.CD007815.pub2. PMID 23728667. http://www.cochrane.org/CD007815/SCHIZ_quetiapine-versus-typical-antipsychotic-drugs-for-schizophrenia.
- ↑ 17.0 17.1 17.2 17.3 17.4 "Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis". Lancet 382 (9896): 951–962. September 2013. doi:10.1016/S0140-6736(13)60733-3. PMID 23810019.
- ↑ 18.0 18.1 "Quetiapine for schizophrenia". The Cochrane Database of Systematic Reviews 2004 (2): CD000967. 2004. doi:10.1002/14651858.CD000967.pub2. PMID 15106155.
- ↑ "Pharmacologic treatment of schizophrenia". Dialogues in Clinical Neuroscience 12 (3): 345–357. 2010. doi:10.31887/DCNS.2010.12.3/jkane. PMID 20954430.
- ↑ "Schizophrenia: a review". American Family Physician 75 (12): 1821–1829. June 2007. PMID 17619525.
- ↑ "Quetiapine versus other atypical antipsychotics for schizophrenia". The Cochrane Database of Systematic Reviews 11 (11): CD006625. November 2013. doi:10.1002/14651858.CD006625.pub3. PMID 24249315.
- ↑ "Adjunctive atypical antipsychotic treatment for major depressive disorder: a meta-analysis of depression, quality of life, and safety outcomes". PLOS Medicine 10 (3): e1001403. 2013. doi:10.1371/journal.pmed.1001403. PMID 23554581.
- ↑ 23.0 23.1 23.2 British National Formulary (BNF) 65. London, UK: Pharmaceutical Press. 2013. p. 235. ISBN 9780857110848. https://books.google.com/books?id=fZLvtHELVdMC.
- ↑ 24.0 24.1 24.2 Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. 2013. ISBN 978-0-9805790-9-3. https://books.google.com/books?id=tn3-swEACAAJ.
- ↑ "Quetiapine and rivastigmine and cognitive decline in Alzheimer's disease: randomised double blind placebo controlled trial". BMJ 330 (7496): 874. April 2005. doi:10.1136/bmj.38369.459988.8F. PMID 15722369.
- ↑ 26.0 26.1 "Safety of low doses of quetiapine when used for insomnia". The Annals of Pharmacotherapy 46 (5): 718–722. May 2012. doi:10.1345/aph.1Q697. PMID 22510671.
- ↑ 27.0 27.1 "Quetiapine for insomnia: A review of the literature". American Journal of Health-System Pharmacy 71 (5): 394–402. March 2014. doi:10.2146/ajhp130221. PMID 24534594.
- ↑ "Quetiapine for primary insomnia: Consider the risks". Cleveland Clinic Journal of Medicine 88 (5): 286–294. May 2021. doi:10.3949/ccjm.88a.20031. PMID 33941603.
- ↑ Off-Label Use of Atypical Antipsychotics: An Update (Report). September 2011. PMID 22132426.
- ↑ Off-Label Use of Atypical Antipsychotics: An Update. Comparative Effectiveness Reviews, No. 43. Rockville: Agency for Healthcare Research and Quality. 2011.
- ↑ "Safety of low doses of quetiapine when used for insomnia". The Annals of Pharmacotherapy 46 (5): 718–722. May 2012. doi:10.1345/aph.1Q697. PMID 22510671.
- ↑ "Comparative effects of pharmacological interventions for the acute and long-term management of insomnia disorder in adults: a systematic review and network meta-analysis" (in English). Lancet 400 (10347): 170–184. July 2022. doi:10.1016/S0140-6736(22)00878-9. PMID 35843245.
- ↑ "Effects of quetiapine on sleep in nonpsychiatric and psychiatric conditions". The Annals of Pharmacotherapy 43 (4): 707–713. April 2009. doi:10.1345/aph.1L320. PMID 19299326.
- ↑ "Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia, predictors of metabolic dysregulation, and association with psychopathology: a systematic review and network meta-analysis". The Lancet. Psychiatry 7 (1): 64–77. January 2020. doi:10.1016/s2215-0366(19)30416-x. PMID 31860457.
- ↑ "Dose-dependent effects of antipsychotics on efficacy and adverse effects in schizophrenia". Behavioural Brain Research 402: 113098. March 2021. doi:10.1016/j.bbr.2020.113098. PMID 33417992.
- ↑ "Use of low-dose quetiapine increases the risk of major adverse cardiovascular events: results from a nationwide active comparator-controlled cohort study". World Psychiatry 21 (3): 444–451. October 2022. doi:10.1002/wps.21010. PMID 36073694.
- ↑ Højlund M (12 September 2022). Low-dose Quetiapine: Utilization and Cardiometabolic Risk (Ph.D. thesis). University of Southern Denmark. doi:10.21996/mr3m-1783.
- ↑ "Quetiapine treatment of children and adolescents with Tourette's disorder". Journal of Child and Adolescent Psychopharmacology 13 (3): 295–299. 2003. doi:10.1089/104454603322572624. PMID 14642017.
- ↑ Oliver Sacks "Musicophilia" Knopf NY 2007 P.67
- ↑ "Treatment of sleep dysfunction and psychiatric disorders". Current Treatment Options in Neurology 8 (5): 367–375. September 2006. doi:10.1007/s11940-006-0026-6. PMID 16901376.
- ↑ Kyle, Kevin; Bronstein, Jeff M. (25 May 2020). "Treatment of psychosis in Parkinson's disease and dementia with Lewy Bodies: A review". Parkinsonism & Related Disorders 75: 55–62. doi:10.1016/j.parkreldis.2020.05.026. ISSN 1873-5126. PMID 32480308. https://pubmed.ncbi.nlm.nih.gov/32480308. "In clinical practice, quetiapine is more readily used than clozapine, given its improved side effect profile compared to clozapine. Despite frequent use in clinical practice, its efficacy in PDP is less clear. ... The quetiapine evidence base is thus ambiguous, lacking consistent support from RCTs in PDP. In DLB there is reliance upon retrospective studies, with the inherent limitations therein. As with clozapine, sedation and orthostasis can be limiting factors. In clinical practice, despite the paucity of consistent evidence, quetiapine has been the first line antipsychotic therapy due to its side effect profile and lower neuroleptic sensitivity risk.".
- ↑ "Quetiapine in the treatment of psychosis in Parkinson's disease". Therapeutic Advances in Neurological Disorders 3 (6): 339–350. November 2010. doi:10.1177/1756285610389656. PMID 21179595.
- ↑ 43.0 43.1 43.2 43.3 The Maudsley prescribing guidelines in psychiatry. West Sussex: Wiley-Blackwell. 2012. ISBN 9780470979693. https://books.google.com/books?id=KY_2Qk4LqVYC.
- ↑ 44.0 44.1 44.2 "PRODUCT INFORMATION STADA(TM) Quetiapine (quetiapine fumarate Tablets 25 mg, 100 mg, 200 mg, 300 mg)". STADA Pharmaceuticals Australia Pty Limited. 30 November 2012. https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2012-PI-03014-1.
- ↑ 45.0 45.1 "Tardive dyskinesia and new antipsychotics". Current Opinion in Psychiatry 21 (2): 151–156. March 2008. doi:10.1097/YCO.0b013e3282f53132. PMID 18332662.
- ↑ "Tardive dyskinesia". Current Treatment Options in Neurology 13 (3): 231–241. June 2011. doi:10.1007/s11940-011-0117-x. PMID 21365202.
- ↑ "Antipsychotic-induced weight gain: a comprehensive research synthesis". The American Journal of Psychiatry 156 (11): 1686–96. November 1999. doi:10.1176/ajp.156.11.1686. PMID 10553730.
- ↑ Seroquel Prescribing Information www.astrazeneca-us.com
- ↑ Joint Formulary Committee BMJ, ed (March 2009). "4.2.1". British National Formulary (57 ed.). United Kingdom: Royal Pharmaceutical Society of Great Britain. p. 192. ISBN 978-0-85369-845-6. "Withdrawal of antipsychotic drugs after long-term therapy should always be gradual and closely monitored to avoid the risk of acute withdrawal syndromes or rapid relapse."
- ↑ 50.0 50.1 50.2 50.3 50.4 (in en) Adverse Syndromes and Psychiatric Drugs: A Clinical Guide. OUP Oxford. 2004. pp. 207–216. ISBN 9780198527480. https://books.google.com/books?id=CWR7DwAAQBAJ&pg=PA207.
- ↑ "Does antipsychotic withdrawal provoke psychosis? Review of the literature on rapid onset psychosis (supersensitivity psychosis) and withdrawal-related relapse". Acta Psychiatrica Scandinavica 114 (1): 3–13. July 2006. doi:10.1111/j.1600-0447.2006.00787.x. PMID 16774655.
- ↑ (in en) Adherence to Antipsychotics in Schizophrenia. Springer Science & Business Media. 2013. p. 85. ISBN 9788847026797. https://books.google.com/books?id=odE-AgAAQBAJ&pg=PA85.
- ↑ "Intentional Recreational Abuse of Quetiapine Compared to Other Second-generation Antipsychotics". The Western Journal of Emergency Medicine 18 (2): 243–250. February 2017. doi:10.5811/westjem.2016.10.32322. PMID 28210359.
- ↑ "Quetiapine Misuse and Abuse: Is it an Atypical Paradigm of Drug Seeking Behavior?". Journal of Research in Pharmacy Practice 6 (1): 12–15. 2017. doi:10.4103/2279-042X.200987. PMID 28331860.
- ↑ "Is There a Potential of Misuse for Quetiapine?: Literature Review and Analysis of the European Medicines Agency/European Medicines Agency Adverse Drug Reactions' Database". Journal of Clinical Psychopharmacology 38 (1): 72–79. February 2018. doi:10.1097/JCP.0000000000000814. PMID 29210868.
- ↑ "Comparison of Quetiapine Abuse and Misuse Reports to the FDA Adverse Event Reporting System With Other Second-Generation Antipsychotics". Substance Abuse 13: 1178221819844205. 2019. doi:10.1177/1178221819844205. PMID 31068753.
- ↑ "Quetiapine Abuse Fourteen Years Later: Where Are We Now? A Systematic Review". Substance Use & Misuse 55 (2): 304–313. 2020. doi:10.1080/10826084.2019.1668013. PMID 31573374.
- ↑ Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. 2008. pp. 1355–1357.
- ↑ "Postmortem femoral blood reference concentrations of aripiprazole, chlorprothixene, and quetiapine". Journal of Analytical Toxicology 39 (1): 41–44. Jan 2015. doi:10.1093/jat/bku121. PMID 25342720.
- ↑ "Postmortem Quetiapine Reference Concentrations in Brain and Blood". Journal of Analytical Toxicology 39 (7): 557–561. September 2015. doi:10.1093/jat/bkv072. PMID 26159868.
- ↑ "PDSP Ki Database". University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. https://pdsp.unc.edu/databases/pdsp.php?knowID=0&kiKey=&receptorDD=&receptor=&speciesDD=&species=&sourcesDD=&source=&hotLigandDD=&hotLigand=&testLigandDD=&testFreeRadio=testFreeRadio&testLigand=quetiapine&referenceDD=&reference=&KiGreater=&KiLess=&kiAllRadio=all&doQuery=Submit+Query.
- ↑ 62.00 62.01 62.02 62.03 62.04 62.05 62.06 62.07 62.08 62.09 62.10 62.11 62.12 62.13 62.14 62.15 62.16 62.17 62.18 62.19 62.20 62.21 62.22 62.23 62.24 62.25 62.26 62.27 62.28 62.29 62.30 62.31 62.32 62.33 62.34 62.35 62.36 62.37 62.38 62.39 62.40 62.41 Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid18059438 - ↑ 63.00 63.01 63.02 63.03 63.04 63.05 63.06 63.07 63.08 63.09 63.10 63.11 63.12 63.13 63.14 63.15 63.16 63.17 63.18 "Risperidone compared with new and reference antipsychotic drugs: in vitro and in vivo receptor binding". Psychopharmacology 124 (1–2): 57–73. March 1996. doi:10.1007/bf02245606. PMID 8935801.
- ↑ AstraZeneca. "Seroquel (quietapine fumarate) tablets". Www1.astrazeneca-us.com. 276521. http://www1.astrazeneca-us.com/pi/Seroquel.pdf.
- ↑ 65.0 65.1 "Binding of antipsychotic drugs to human brain receptors focus on newer generation compounds". Life Sciences 68 (1): 29–39. November 2000. doi:10.1016/S0024-3205(00)00911-5. PMID 11132243.
- ↑ "Therapeutics of schizophrenia". Neuropsychopharmacology: the fifth generation of progress. American College of Neuropsychopharmacology. 2002. ISBN 978-0-7817-2837-9. https://books.google.com/books?id=BKwkonZwZD0C&pg=PA778.
- ↑ "Seroquel Official FDA information, side effects and uses". Drugs.com. https://www.drugs.com/pro/seroquel.html.
- ↑ AstraZeneca Pharmaceuticals LP (March 2011). "SEROQUEL (quetiapine fumarate) tablet, extended release". National Library of Medicine. Section 12.2: Pharmacodynamics. http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=41375#section-15.2.
- ↑ National Institute of Mental Health. PDSD Ki Database (Internet) [cited 2013 Sep 18]. Chapel Hill (NC): University of North Carolina. 1998-2013. Available from: "...list shows the top 100 Receptors/Targets in Ki". http://pdsp.med.unc.edu/pdsp.php.
- ↑ "N-desalkylquetiapine, a potent norepinephrine reuptake inhibitor and partial 5-HT1A agonist, as a putative mediator of quetiapine's antidepressant activity". Neuropsychopharmacology 33 (10): 2303–2312. September 2008. doi:10.1038/sj.npp.1301646. PMID 18059438.
- ↑ "Active metabolites as antidepressant drugs: the role of norquetiapine in the mechanism of action of quetiapine in the treatment of mood disorders". Frontiers in Psychiatry 4: 102. September 2013. doi:10.3389/fpsyt.2013.00102. PMID 24062697.
- ↑ "Anticholinergic activity of 107 medications commonly used by older adults". Journal of the American Geriatrics Society 56 (7): 1333–1341. July 2008. doi:10.1111/j.1532-5415.2008.01737.x. PMID 18510583.
- ↑ "Mechanism of action of quetiapine". Psychopharmacology Institute. http://psychopharmacologyinstitute.com/antipsychotics/quetiapine/mechanism-of-action/.
- ↑ "Does fast dissociation from the dopamine d(2) receptor explain the action of atypical antipsychotics?: A new hypothesis". The American Journal of Psychiatry 158 (3): 360–369. March 2001. doi:10.1176/appi.ajp.158.3.360. PMID 11229973.
- ↑ "Atypical antipsychotics: mechanism of action". Canadian Journal of Psychiatry. Revue Canadienne de Psychiatrie 47 (1): 27–38. February 2002. doi:10.1177/070674370204700106. PMID 11873706.
- ↑ "D(2) and 5HT(2A) receptor occupancy of different doses of quetiapine in schizophrenia: a PET study". European Neuropsychopharmacology 11 (2): 105–110. April 2001. doi:10.1016/S0924-977X(00)00133-4. PMID 11313155.
- ↑ "Drug Use Evaluation: Low Dose Quetiapine (Seroquel/Seroquel XR)". Oregon State. http://www.govexec.com/pdfs/013111bb1a.pdf.
- ↑ 78.0 78.1 "Quetiapine". https://www.drugbank.ca/drugs/DB01224.
- ↑ "Seroquel". https://www.accessdata.fda.gov/drugsatfda_docs/label/2004/20639se1-017,016_seroquel_lbl.pdf.
- ↑ "Quetiapine: preclinical studies, pharmacokinetics, drug interactions, and dosing". The Journal of Clinical Psychiatry 63 (Suppl 13): 5–11. 2002. PMID 12562141.
- ↑ "Review of quetiapine and its clinical applications in schizophrenia". Expert Opinion on Pharmacotherapy 1 (4): 783–801. May 2000. doi:10.1517/14656566.1.4.783. PMID 11249516.
- ↑ 82.0 82.1 82.2 82.3 "Seroquel". https://www.accessdata.fda.gov/drugsatfda_docs/label/2004/20639se1-017,016_seroquel_lbl.pdf.
- ↑ Warawa, E. J.; Migler, B. M.; 1988, U.S. Patent 4,879,288.
- ↑ "AstraZeneca Submits an NDA For Sustained Release Formulation Seroquel XR. For the treatment of schizophrenia" (Press release). AstraZeneca. 18 July 2006. Archived from the original on 5 November 2006. Retrieved 1 January 2007.
- ↑ "AstraZeneca Submits EU and Canadian Regulatory Filings for Sustained Release Formulation SEROQUEL XR for the Treatment of Schizophrenia" (Press release). AstraZeneca. 19 October 2006. Archived from the original on 7 November 2006. Retrieved 1 January 2007.
- ↑ "FDA Approves AstraZeneca's Once-Daily SEROQUEL XR Extended-Release Tablets For The Treatment Of Schizophrenia" (Press release). AstraZeneca. 18 May 2007. Archived from the original on 28 September 2007. Retrieved 2 August 2007.
- ↑ "Second Quarter and Half Year Results 2007" (Press release). AstraZeneca. 26 July 2007. Archived from the original on 24 August 2007. Retrieved 2 August 2007.
- ↑ "Seroquel XR Receives Approval from FDA for Maintenance Treatment of Schizophrenia" (Press release). AstraZeneca. 16 November 2007. Archived from the original on 4 December 2007. Retrieved 3 December 2007.
- ↑ Notice of Compliance Information - Seroquel XR[yes|permanent dead link|dead link}}] 27 September 2007, retrieved 3 December 2007
- ↑ "Biovail Announces Filing of ANDA for Quetiapine XR Tablets" (Press release). Biovail. 1 December 2008. Archived from the original on 9 August 2007.
- ↑ "AstraZeneca Receives FDA Complete Response Letter on Seroquel XR for Major Depressive Disorder" (Press release). AstraZeneca. 24 December 2008. Archived from the original on 26 October 2010. Retrieved 28 December 2008.
- ↑ 92.0 92.1 92.2 "Pharmaceutical Giant AstraZeneca to Pay $520 Million for Off-label Drug Marketing". Justice news, US Department of Justice. 27 April 2010. http://www.justice.gov/opa/pr/2010/April/10-civ-487.html.
- ↑ "Quetiapine Indications: FDA-Approved and Off-label Uses". Psychopharmacology Institute. http://psychopharmacologyinstitute.com/antipsychotics/quetiapine/quetiapine-indications/.
- ↑ "QUETIAPINE FUMARATE". Food and Drug Administration. April 2007. http://www.accessdata.fda.gov/scripts/cder/ob/docs/obdetail.cfm?Appl_No=020639&TABLE1=OB_Rx.
- ↑ "AstraZeneca Receives FDA Approval for SEROQUEL in Bipolar Mania" (Press release). AstraZeneca. 13 January 2004. Archived from the original on 8 June 2011.
- ↑ "Pioneering science, life-changing medicines". 2013. https://www.sec.gov/Archives/edgar/data/901832/000095010314001994/dp44853_ex1501.htm.
- ↑ "Seroquel suit claims 'so much' is poured into marketing and away from research". The Madison / St. Clair Record. 2 August 2007. http://www.madisonrecord.com/news/198781-seroquel-suit-claims-so-much-is-poured-into-marketing-and-away-from-research.
- ↑ "AstraZeneca May Link Seroquel, Diabetes, Doctor Says". Bloomberg.com (Bloomberg L.P.). 11 March 2009. https://www.bloomberg.com/apps/news?pid=newsarchive&sid=ayzJsK2HlF6s.
- ↑ "AstraZeneca wins bellwether Seroquel case". FiercePharma. 19 March 2010. http://www.fiercepharma.com/story/astrazeneca-wins-bellwether-seroquel-case/2010-03-19.
- ↑ "AstraZeneca pays out million dollar damages". The Local. 9 August 2010. http://www.thelocal.se/28260/20100809/.
- ↑ "Questions loom over drug for sleepless vets - Marine Corps News | News from Afghanistan & Iraq". Marine Corps Times. http://www.marinecorpstimes.com/news/2010/08/ap-veterans-affairs-investigation-into-seroquel-083010/.
- ↑ "Heart Warning Added to Label on Popular Antipsychotic Drug". NyTimes. 19 July 2011. https://www.nytimes.com/2011/07/19/health/19drug.html?_r=2.
- ↑ "The Deadly Corruption of Clinical Trials". Mother Jones. September–October 2010. https://www.motherjones.com/environment/2010/09/dan-markingson-drug-trial-astrazeneca.
- ↑ "Minnesota bioethicists critique their university". Science. 7 December 2010. http://news.sciencemag.org/scienceinsider/2010/12/minnesota-bioethicists-critique-.html.
- ↑ "Press release, 25 August 2011" (Press release). MHRA. Archived from the original on 19 March 2012. Retrieved 9 July 2012.
- ↑ "Drug Alerts". MHRA. http://www.mhra.gov.uk/Publications/Safetywarnings/DrugAlerts/CON126268.
- ↑ "Nurofen Plus tampering: Christopher McGuire jailed". BBC News. 28 May 2012. https://www.bbc.co.uk/news/uk-england-london-18203634.
External links
 |